Workflow Intravenous Therapy Kickstart
|
This page is currently under construction. |
Inhoud
- 1 Introduction
- 2 Prescribe
- 3 Dispense
- 4 Administer
- 5 Practical Examples of Infusions
- 5.1 Practical example 1: Preparation of infusion by pharmacist
- 5.2 Practical Example 2: Administration with a flushing agent other than the solvent
- 5.3 Practical Example 3: Administration with the Same Flushing Agent as the Solvent
- 5.4 Practical Example 4: Registering an incomplete infusion administration by means of a correction
- 5.5 Practical Example 5: Application of a correction method in the registration of MTD due to a change in the administration rate
1 Introduction
The information standard for Medication Process 9 (MP9) has not yet been fully developed. Further development is needed, particularly for the exchange of data relating to infusion therapy. Specific agreements have been made for the Kickstart based on decision VO37 of the Core Team. However, this decision does not sufficiently clarify what infusion data is and is not exchanged. Further development has been achieved in collaboration with the administration sectors, Medical Specialist Care and various software suppliers. This is explained below in various situations.
This approach applies to the Kickstart and will be further developed simultaneously.
2 Prescribe
The following agreement applies to the prescription of infusion therapy in the Kickstart:
The active substance is stated in the medication agreement (MA). A reference to a schedule or protocol indicating how the infusion should be prepared, as well as the solvent, can be included in the 'Explanation' data element. This also applies to (short-acting) insulins.
If multiple active substances are prescribed, this usually results in the same number of MAs or a magistral. In the case of an infusion preparation that is included in the G-standard and contains multiple active substances, that specific product can be selected in the MA. This does not result in multiple MAs.
3 Dispense
3.1 Clinical situation
In a clinical situation, the solvent is usually not prescribed separately. A single administration agreement (TA) containing the active substance is typically sufficient.
3.2 Ambulatory Situation
In an ambulatory situation, both the pharmacy and the administrator are able to prepare the medicine for administration.
3.2.1 Preparation of infusion by pharmacist
The pharmacist prepares a TA with compounded medication, in which both the drug and the solvent are registered as ingredient. This results in a single entry on the administration list. See practical example 1 for details.
3.2.2 Preparation of infusion by administrator
When the infusion is prepared by the administrator, the pharmacist does not only dispense the medicine. Associated aids are also provided, such as a solvent and an agent for flushing the line. Therefore, two separate, parallel TAs and medication dispensings (MVEs) are created under one pharmaceutical treatment (MBH): one for the medicine and one for the solvent. This results in two separate lines on the administration list. This must also clearly state what the aid is intended to be used for. See practical examples 1 and 2 for details.
4 Administer
Specific agreements have been established for medication administrations (MTDs) that only apply during the Kickstart. Not all MTDs and other infusion data will be exchanged and are therefore not available in the chain, for example, for an administration overview. Below are the details:
- Single active substance: MTDs of infusions with a single active substance, whether or not prepared for administration, are recorded in the information system and exchanged. Associated MAs and TAs are also exchanged.
- Dual active substances: MTDs of infusions with two active substances can be registered but are not exchanged. Unless it is registered as a magistral in one MA and TA. In that case, the MTDs can be exchanged. Associated MAs and TAs are also exchanged here.
- Infusion rate: The infusion rates described in the MA and TA are also included in the MTD and can be exchanged. This applies to both continuous infusions and bolus doses. See practical example 5 for more information.
However, if infusion rates are not specified in the MA or TA and an administrator makes a change themselves, this is not exchanged in the MDT, for example in the case of a perfuser pump.
- Volume: When attaching a new syringe pump or infusion bag to the patient, the total volume is registered in the MTD and exchanged. If only part of the total volume has been administered, this can be updated by correcting the MTD. This is done by creating an additional MTD, in which the remaining volume of the infusion bag is registered. This is recorded as a negative number, for example -200 ml. With this, the total administration can be calculated. The original volume of for example 1 litre, minus the residual volume of 200 ml, results in a total administration of 1000 – 200 = 800 ml. See practical example 4 for details.
- Admission/discharge: In situations of admission or discharge, it may be necessary to provide a written transfer of MTDs or other infusion data that cannot be exchanged. Relevant MTDs from the past 24 hours can be provided. In addition, the volume of an infusion that is being taken along can also be stated.
5 Practical Examples of Infusions
5.1 Practical example 1: Preparation of infusion by pharmacist
The prescriber issues a prescription for ceftriaxone 2000 mg, to be administered once daily for a duration of six weeks. In the notes of the MA the prescriber specifies that the ceftriaxone must be delivered using an elastomeric pump.
The pharmacy prepares the 2000 mg dose of ceftriaxone in the elastomeric pump and creates a TA for a compounded medication that includes both the active substance and the solvent.
If the elastomeric pump is registered as a medical device from the G-Standard (Dutch drug database) in the TA, it will be made available accordingly. It is up to the receiving system to ensure this TA is not displayed in the medication overview.
A healthcare professional records the administration of the ceftriaxone when the pump is connected.
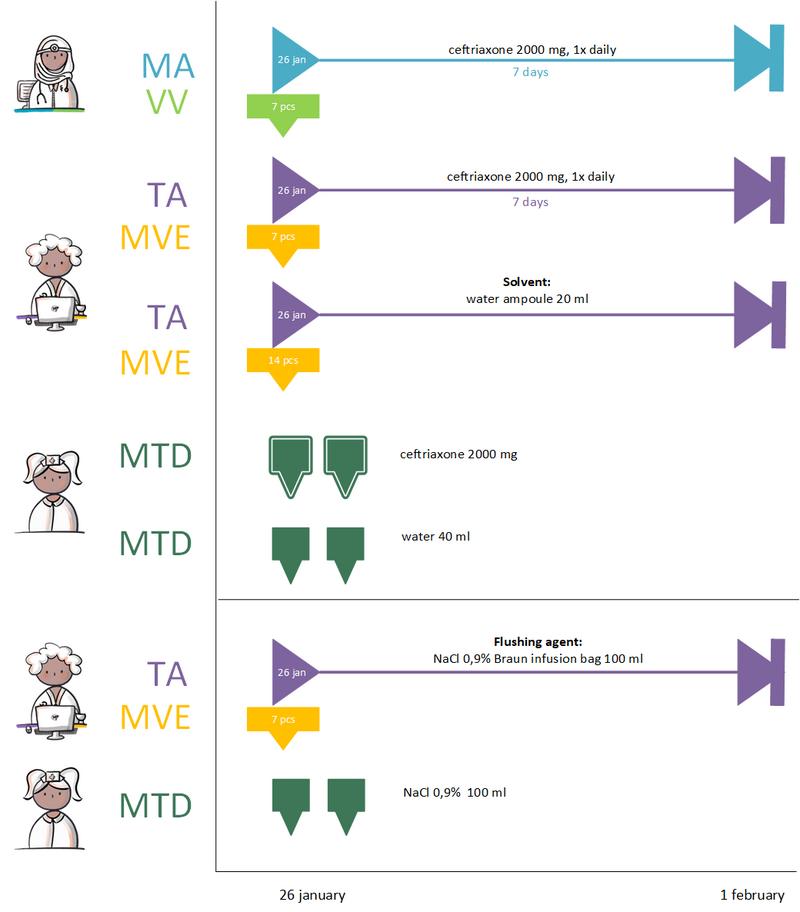
5.2 Practical Example 2: Administration with a flushing agent other than the solvent
Ceftriaxone 2000 mg intravenously has been prescribed for at least 7 days. The patient is receiving this treatment at home through home care using a PICC line, a central venous catheter suitable for extended use. The line must be flushed daily after the administration of the antibiotic. The prescriber creates an MA and VV for the active substance, ceftriaxone.
The pharmacist establishes two TAs for medication administration: one for the antibiotic and one for the solvent, Water for Injections. Additionally, a third TA is created for 0.9% NaCl infusion bags used for saline flushes, categorized under a separate MBH. Each of these provisions is recorded in its respective MVE entry.
While the administrations of the antibiotic and solvent can be grouped together on the administration list under the same MBH. The flushing agent may remain separate due to its association with a different MBH. Consequently, three distinct MTDs will be generated.
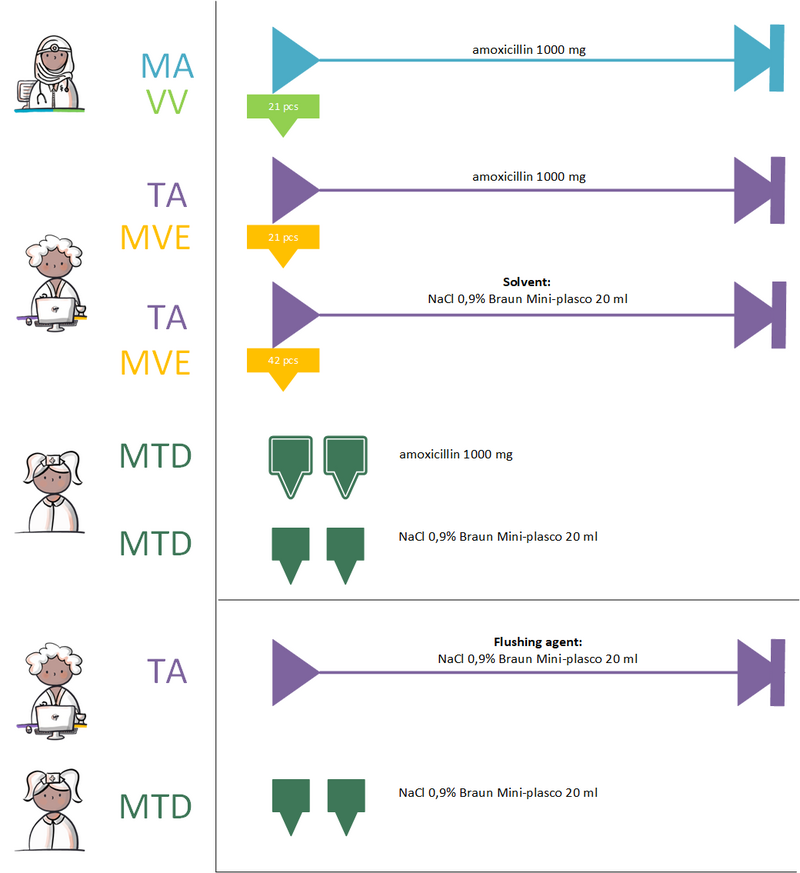
5.3 Practical Example 3: Administration with the Same Flushing Agent as the Solvent
Due to pneumonia, the patient is prescribed an intravenous course of antibiotics. The administration will be prepared at the bedside.
The prescriber issues a MA for amoxicillin 1000 mg.
The pharmacy, under this MBH, prepares two TA's for the amoxicillin powder for injection, as well as for the NaCl 0.9% ampoules used to dilute the amoxicillin. The annotation specifies that these are to be used for the preparation of amoxicillin prior to administration.
NaCl 0.9% ampoules are required both for dissolving the amoxicillin and for flushing the intravenous line. A separate TA must be created for flushing the line with NaCl 0.9%. Since only one dispensing of a product per day is allowed, the dispensed quantity of NaCl 0.9% ampoules must be sufficient for both purposes: preparing the antibiotic for administration and flushing the IV line.
The two administrations—the antibiotic and its solvent—can be grouped under the MBH, allowing them to be easily grouped together on the administration list. The flushing solution may not be grouped together due to a different MBH. This results in three separate MTDs.
5.4 Practical Example 4: Registering an incomplete infusion administration by means of a correction
A healthcare provider prescribes a medication order for morphine 1mg/ml, with a total volume of 100 ml for administration per infusion. The prescriber sets the administration rate at 10 mg/hour. The pharmacist prepares a medication order for Sendolor 1 mg/ml with an administration rate of 10 mg/hour. During administration, after 1 hour, patient gets discomfort from the infusion and medication through the infusion is stopped. After one hour there is 90 mg of medication in the bag. This amount is recorded as -90mg, indicating that 10 mg has been administered (100-90=10 mg). For similar correction examples, please refer to section 4.3.15 of the FO.
5.5 Practical Example 5: Application of a correction method in the registration of MTD due to a change in the administration rate
A prescriber makes a MA for morphine 1 mg/ml administration by infusion. The administration rate is set by the prescriber at 0.5 mg/hour. The pharmacist makes a TA for Sendolor 1 mg/ml with administration rate 0.5 mg/hour.
The caregiver who connects the Sendolor to the patient records the medication administration.
After three days, the pump rate is increased to 1 mg/hour by the prescriber. A change in the rate cannot be exchanged in the MTD. This is solved by a correction: The caregiver corrects the medication administration, by registering in a new MTD the remaining amount as a negative number, in this case -62 mg. This allows the calculation that on balance 100 - 62 = 38 mg was given at this administration rate.
Then, in a subsequent MTD, the new run rate is recorded with the remaining amount, in this case 62 mg.