Practical examples functional design MP9
Practical examples are described for the various subprocesses. The practical situations are in many cases derived from general practice but are illustrative for comparable situations in another setting. Several of these concrete practical situations include a visualisation. This visualisation shows the healthcare professionals and building blocks involved in the situation. Changes in building blocks or new building blocks are shown over time.
Practical examples Medication verification
Practical examples Prescribe
Practical examples Dispense
Practical examples Administer
Practical examples Use
Practical examples GDS
back to FD
back to page with additional documentation
Practical examples
Inhoud
- 1 Medication for a definite period
- 2 Medication for an indefinite period
- 3 ‘As needed’ medication
- 4 Course of treatment when necessary, with uncertain startDateTime
- 5 Medication for single use with specific intake time
- 6 Dosing with minimum interval between doses
- 7 Two doses of the same medicine at the same time
- 8 The same medication with different strengths at the same time
- 9 New medication agreement, no dispense request
- 10 Dosage reduction, no dispense request
- 11 Dosage increase, no dispense request
- 12 New dispense request for existing medication agreement from another prescriber
- 13 Dispense request with number of repeats
- 14 Do not dispense before a certain date
- 15 Prescription no longer required after first dispense request
- 16 Discontinuing medication
- 17 Stopping medication without medication agreement
- 18 Interrupting and resuming medication
- 19 Interrupting medication because of a procedure
- 20 Canceling a previously sent prescription
- 21 Modifying a medication agreement by another prescriber
- 22 Changing someone else’s medication agreement by a non-prescriber with delegated responsibility
- 23 Starting medication before admission
- 24 Parallel medication agreements after failure of the prescriber’s information system
- 25 Two medication agreements in different MBHs for the same medication
- 26 Merging building blocks under a single pharmaceutical treatment
- 27 Sending renal function value along with new prescription
- 28 Renal function value reason for dosage change
- 29 Renal function test required due to type of medicine
- 30 Starting a variable dosing regimen
- 31 Interim modification of a variable dosing regimen
- 32 Stopping medication with a variable dosing regimen
- 33 Paper prescription – no more allowed
- 34 Own items (90 million numbers)
- 35 New medication agreement - dispensing the same product
- 36 New medication agreement – different administration agreement
- 37 Proposal medication agreement sent to prescriber for dose adjustment
- 38 Proposal medication agreement sent to prescriber for temporarily stopping medication
- 39 Proposal medication agreement sent to prescriber for adding another medicine
- 40 Long-term medication with repeats in the dispense request
- 41 Long-term medication, supplier makes partial dispensings
- 42 RequestDate with multiple partial supplies
- 43 Patient requests repeat prescription via prescriber
- 44 Patient requests repeat prescription via supplier
- 45 Proactive repeat prescription by supplier
- 46 Dispensing a larger quantity than prescribed
- 47 Dispensing a smaller quantity than prescribed
- 48 Starting medication in GDS
- 49 Adding medication to GDS
- 50 GDS supplier delivers a different trade product
- 51 Supplier changes brand product
- 52 Parallel administration agreements for GDS and non-GDS dispensing
- 53 Parallel administration agreements, one of which is modified
- 54 Dispensing under someone else’s administration agreement
- 55 Modifying another person’s administration agreement
- 56 Increasing the dose of GDS medication in a new MBH
- 57 Increasing the dose of GDS medication within the same MBH
- 58 Increasing the dose of GDS medication with different administration times
- 59 Reducing the dose of GDS medication in a new MBH
- 60 Reducing the dose of GDS medication within the same MBH
- 61 Changing GDS medication to ‘as needed’ medication
- 62 Changing GDS medication to partly GDS and partly ‘as needed’ medication
- 63 Administering a changed tablet quantity
- 64 Correcting an administration
- 65 Correcting administration after the patient spits out a tablet
- 66 Medication not administered
- 67 Postponing an administration
- 68 Recording separate MTDs per InjectionPatchSite
- 69 Reducing dosage on the patient’s initiative
- 70 Stopping medication on the patient’s initiative
- 71 Patient requests repeat medication from the prescriber via proposal dispense request
- 72 Medication is used as agreed
- 73 Medication is used as agreed and patients intends to keep doing so
- 74 Medication is used but not as agreed
- 75 Medication not used, this is not as agreed
- 76 Medication is not used, this is as agreed
- 77 Medication is not used after the PeriodOfUse and this is as agreed
- 78 Medication is not used during the PeriodOfUse and this is as agreed
- 79 Medication is used but unknown whether this is as agreed
- 80 Correcting inadvertently outstanding medication (‘orphan’) during medication verification
- 81 Medication for single use without a specific intake time
- 82 DurationOfUse in the MVE for non-daily GDS medication
- 83 DurationOfUse in the MVE when bridging the period until medication is included in GDS
- 84 Dispensing after approval of proposal medication agreement for additional medication
- 85 Dispensing before approval of proposal medication agreement for additional medication
- 86 Dispensing before approval of proposal medication agreement for replacement medication
- 87 Document history
1 Medication for a definite period
The GP prescribes a course of treatment to a patient with a urinary tract infection. On 27 January, the following is agreed :
- MA: nitrofurantoin 100 mg MR capsule, 1 capsule twice daily, from today, for 5 days
She records this MA in the information system and also records a VV:
- VV: nitrofurantoin 100 mg MR capsule; 10 capsules
2 Medication for an indefinite period
The GP prescribes a diuretic for an indefinite period to a patient with hypertension. On 30 March, the following is agreed:
- MA: hydrochlorothiazide 12.5 mg tablet, 1 tablet once daily, from today
The GP leaves the endDateTime open, making it clear that this is an MA for an indefinite period. In addition, he records a VV:
- VV: hydrochlorothiazide 12.5 mg tablet; 90 tablets
3 ‘As needed’ medication
The GP has diagnosed a patient with migraine and prescribes rizatriptan for this condition:
- MA: rizatriptan 10 mg tablet, 1 tablet SL once daily, as needed
In the AsNeeded.Condition data element of the MA the GP records ‘Take first tablet at next clear migraine attack’.
The GP also records a VV:
- VV: rizatriptan 10 mg orodispersible tablet; 6 tablets
4 Course of treatment when necessary, with uncertain startDateTime
A patient occasionally suffers from erysipelas as a result of thrombosis of the leg. Erysipelas is a serious infection that must be treated as quickly as possible. The GP therefore prescribes a course of antibiotics that the patient can start immediately as soon as the erysipelas recurs:
- MA: flucloxacillin 500 mg capsule, 1 capsule 4 times daily, for 10 days
In the data element PeriodOfUse.Condition of the MA, she records: ‘Start on recurrence of erysipelas’
The GP also records a VV:
- VV: flucloxacillin 500 mg capsule; 40 capsules
After the patient has had an episode of erysipelas, the PeriodOfUse is known. The GP records a stop-MA containing the actual startDateTime or endDateTime. She can then create a new MA for a possible subsequent episode of erysipelas.

5 Medication for single use with specific intake time
A patient has to undergo surgery in hospital and is worried about this. The GP decides to prescribe a single dose of diazepam for use on the evening before the operation. She indicates this by entering a startDateTime and endDateTime in the PeriodOfUse.

6 Dosing with minimum interval between doses
A patient with cancer is in a lot of pain. The specialist prescribes:
- MA: oxycodone 5 mg tablet, as needed, with a maximum of 1 tablet 4 times daily, leave at least 4 hours between doses
The instructions ‘as necessary’ and ‘with a maximum of 1 tablet 4 times daily’ are recorded in the standard way in the InstructionsForUse. The instruction ‘leave at least 4 hours between doses’ is recorded in free text in AdditionalInstructions.

7 Two doses of the same medicine at the same time
A patient with metastatic prostate cancer is in a lot of pain. On 9 October, the specialist prescribes:
- MA: oxycodone 10 mg tablet, 1 tablet 4 times daily and, as needed, 1 tablet once daily for pain at night
The specialist records this in a single MA with two dosage instructions (in InstructionsForUse.DosingInstructions) and records a VV:
- VV: oxycodone 10 mg tablet; 60 tablets
8 The same medication with different strengths at the same time
On 9 October, a patient with metastatic prostate cancer was prescribed oxycodone 10 mg tablets for pain relief.
- MA 9 October: oxycodone 10 mg tablet, 1 tablet 4 times daily and, as needed, 1 tablet once daily for pain at night
The patient is experiencing increased pain. The specialist therefore stops this MA on 16 October and now prescribes:
- MA 16 October: oxycodone 40 mg MR tablet, 1 tablet twice daily
- MA 16 October: oxycodone 20 mg tablet, as needed, 1 tablet once daily for pain at night
She records this in two MAs, in two new MBHs, as these are different medicines at PRK level, and also differ from the previously prescribed tablets. She records a VV for each MA.

9 New medication agreement, no dispense request
A man visits his GP with back pain. The symptoms have been present for 3 weeks and he has been taking paracetamol for this. On 30 January, the GP prescribes additional diclofenac:
- MA: diclofenac 50 mg tablet, 1 tablet 3 times daily, from today, for 3 weeks
The patient indicates that he has sufficient stock at home from a previous condition. Therefore, no VV is necessary.

10 Dosage reduction, no dispense request
On 1 January, the GP prescribes tramadol for an indefinite period to his patient because of pain. He also records a VV for 3 months.
- MA: tramadol 50 mg capsule, 1 capsule 4 times daily, from 1 January
- VV: tramadol 50 mg capsule; 360 capsules
The patient may reduce the medication at his own discretion. During a subsequent consultation on 1 February, the patient indicates that for the past week, 1 capsule 3 times daily has been sufficient. The GP modifies the medication by recording a new MA with startDateTime 1 February, along with a technical stop-MA.
- MA: tramadol 50 mg capsule, 1 capsule 3 times daily, from 1 February
A new VV is not immediately required, as the patient still has sufficient stock.

11 Dosage increase, no dispense request
A patient with asthma is prescribed beclomethasone on 10 June.
- MA 10 June: beclomethasone 100 micrograms/dose inhaler, 1 inhalation twice daily, from 10 June
The patient returns to the pulmonologist for a check-up on 8 July. The pulmonologist finds that the asthma is not sufficiently controlled. She changes the medication by recording a new MA with the increased dosage. A technical stop-MA is also recorded.
- MA 8 July: beclomethasone 100 micrograms/dose inhaler, 2 inhalations twice daily, from 8 July
She notes ‘Lack of drug action’ as the reason in the data element ReasonModificationOrDiscontinuation. The patient still has sufficient stock, so a VV is not immediately necessary. The prescription therefore only contains an MA.

12 New dispense request for existing medication agreement from another prescriber
The internist has diagnosed a patient with hypothyroidism and prescribes levothyroxine to her. After some time, she is well adjusted to a dosage of 125 micrograms per day. The internist records this in an MA.
- MA: levothyroxine 125 micrograms tablet, 1 tablet once daily
They agree that the patient will return to the internist for a check-up in a year’s time. In the meantime, the GP will take care of the repeat medication. For this, the GP records a VV and sends it along with the specialist’s MA to the supplier.

13 Dispense request with number of repeats
The prescriber records a VV for an MA in which she fills in the data element NumberOfRefills. In doing so, the prescriber indicates that the supplier may make additional dispenses.
The prescriber can specify in two ways how much of the medicine the supplier may ultimately deliver:
The number of repetitions in combination with the quantity to be dispensed results in:
- (
NumberOfRefills+ 1) xAmount.
In the case of ‘NumberOfRefills = 3’ and ‘Amount = 30 units’, the supplier may dispense 4 times 30 units (a total of 120 units).

Alternatively, the duration of the validity period may be used. In that case, the quantity to be dispensed depends on the dosage recorded in the MA, and can be computed with: (NumberOfRefills + 1) x ValidityPeriod.Duration x Frequency x Dose.
In the case of ‘Dosage = 2 units, 3 times daily’, ‘NumberOfRefills’ = 3, and ‘ValidityPeriod.Duration = 30 days’, the supplier may dispense 4 times (6 units per day x 30 days = 180 units) = a total of 720 units.

14 Do not dispense before a certain date
A patient uses controlled-release oxycodone for chronic pain. Treatment dosage is stable. Due to the risk of misuse, it has been agreed that the medicine will only be dispensed at agreed times.
On 14 April, the GP makes a new VV and wants to specify that oxycodone may only be dispensed from 17 April onwards. Because this cannot be specified in a structured manner in the VV, this instruction is included in free text in the Comment.

15 Prescription no longer required after first dispense request
A 16-year-old woman reports that she wishes to use contraception. After receiving an explanation, she chooses the contraceptive pill. The doctor prescribes Microgynon 20 and explains that she must take one tablet once daily for 21 days, then stop for 7 days, and then repeat the cycle.
- MA: Microgynon 20, 1 tablet once daily for 21 days, stop for 7 days, repeat cycle
- VV: Microgynon 20; 63 tablets
He tells her that if she has no complaints, she can continue to obtain the pill from the pharmacy. No new VV is required for this. This medicine falls under one of the three categories (insulins, oral contraceptive pills and non-oral UR contraceptives) for which prescriptions may be issued for longer than one year. For these categories, it does not matter how the initial VV was completed.
Three months later, the woman asks the pharmacy for a new supply. The pharmacy dispenses the medicine and sends a message that the prescription has been processed to the prescriber.

16 Discontinuing medication
A patient visits the pulmonologist on 13 August for a check-up on his asthma. The patient is experiencing a side effect. He is currently using beclomethasone in accordance with the MA of 10 June.
- MA 10 June: beclomethasone 100 micrograms/dose inhaler, 1 inhalation twice daily, from 10 June
The pulmonologist agrees with the patient to stop this medication and records a stop-MA:
- stop-MA 13 August: beclomethasone 100 micrograms/dose inhaler;
MedicationAgreementStopType‘Discontinued’;endDateTime13 August;ReasonModificationOrDiscontinuation‘Adverse reaction to drug’.
The stop-MA is sent to the supplier and made available to fellow health professionals and the patient.

17 Stopping medication without medication agreement
During his holiday, a patient was prescribed diclofenac for back pain on 13 July. He recorded an MGB for this in his PGO. As there is no digital MA, the MBH started with this MGB.
After returning home, he visits his GP on 28 July because he does not know how long he can continue to use this medicine. During the consultation the GP agrees with the patient to stop the diclofenac. The GP creates a stop-MA within the MBH that was started with the MGB.

18 Interrupting and resuming medication
A patient is being treated with the cholesterol-lowering drug simvastatin.
- MA: simvastatin 40 mg tablet, 1 tablet once daily
On 7 July, she visits her GP, who prescribes clarithromycin as an antibiotic for one week to treat an infection. Because this interacts with simvastatin, simvastatin is interrupted during that week and then resumed.
- stop-MA: simvastatin 40 mg tablet, 1 tablet once daily;
MedicationAgreementStopType‘Suspended’;ReasonModificationOrDiscontinuation'Drug interaction' - MA: clarithromycin 250 mg tablet, 1 tablet twice daily, from 7 July for 1 week
- future MA: simvastatin 40 mg tablet, 1 tablet once daily, from 14 July
19 Interrupting medication because of a procedure
A 62-year-old man takes acetylsalicylic acid 80 mg 1 tablet once daily for coronary artery disease. He needs to undergo surgery to have an intestinal polyp removed. The GP discusses with him that he must stop taking acetylsalicylic acid one week before the procedure and resume taking it the day after the procedure. The GP creates a stop-MA with stop type ‘Suspended’ and a new MA to resume the medication after the procedure.
The date of the procedure is not yet known. In the data element PeriodOfUse.Condition of the stop-MA, the GP therefore indicates that the medication must be stopped one week before the procedure. In the new MA, this data element indicates that the medication must be resumed one day after admission.
As soon as the date of the procedure is known, the MAs will be changed to the actual start and stop dates.

20 Canceling a previously sent prescription
A patient visits his GP in the morning with stomach complaints. The GP prescribes pantoprazole and sends the prescription (MA + VV1) to the GP's preferred pharmacy. The patient calls in the afternoon. He forgot to mention in the morning that he has moved and therefore want to collect the medication from his new pharmacy.
The GP sends the prescription to the first pharmacy again, this time with the VV containing the CanceledIndicator (MA + VV1 ‘canceled’). This informs the supplier that she mustn’t dispense this prescription.
The prescriber then sends a new prescription (MA + VV2) to the patient's new pharmacy.

The corresponding transactions are the following:

21 Modifying a medication agreement by another prescriber
On 10 June, the GP prescribes lisinopril to a patient for hypertension.
- MA 10 June: lisinopril 20 mg tablet, 2 tablets once daily
On 13 August, the patient is admitted to the Accident and Emergency department following a fall due to dizziness. The specialist diagnoses hypotension and decides to reduce the dose of lisinopril. The specialist records a new MA with the lower dosage. In addition, a technical stop-MA is created.
- MA 13 August: lisinopril 20 mg tablet, 1 tablet once daily
The specialist sends the stop-MA and the new MA to the original prescriber.

22 Changing someone else’s medication agreement by a non-prescriber with delegated responsibility
The medical specialist prescribes niraparib to a patient with cancer.
- MA: niraparib 100 mg tablet, 3 tablets once daily
Prior to the planned first dose, the hospital pharmacist assesses the lab values and decides that the dosage should be adjusted to 2 tablets once daily. In the hospital it has been agreed that the hospital pharmacist has delegated prescribing authority (see section 5.2). The hospital pharmacist changes the MA of the medical specialist.
- MA: niraparib 100 mg tablet, 2 tablets once daily
The medical specialist remains the Prescriber of the MA, because the hospital pharmacist is acting on her behalf.

23 Starting medication before admission
For cataract surgery, a patient must start using nepafenac eye drops three days before the procedure. These must be used until three weeks after the operation.
- MA: nepafenac 3 mg/ml eye drops, 1 drop once daily in the morning, for 24 days
If the date of the operation is unknown or uncertain, the data element PeriodOfUse.Condition of the MA indicates that treatment should be started 3 days before the operation.

24 Parallel medication agreements after failure of the prescriber’s information system
The GP prescribes a third-class corticosteroid to a patient with constitutional eczema.
- MA: mometasone 0.1% cream, apply once daily
This is not available, so the supplier changes it to desoximetasone cream.
- TA: desoximetasone 0.25% cream, apply once daily
The GP refers the patient to a dermatologist. The dermatologist wants to change the therapy, but the GP's information system is down. The dermatologist therefore only has access to the supplier's TA and creates a stop-MA and a new MA based on that information.
- MA: desoximetasone 0.25% cream, apply twice daily
This results in two parallel MAs with different PRKs in the same MBH.
Without intervention, these two MAs will continue to be active indefinitely. After a failure, an information system must therefore check whether there have been any changes. Any parallel MA must then be stopped.

After the downtime, the IS reports which MBHs have undergone changes. The GP sees that two parallel MAs have been created. She stops her MA for mometasone cream.
25 Two medication agreements in different MBHs for the same medication
A patient with heart failure and multiple chronic conditions uses various medicines, including diuretics, beta blockers and anticoagulants. During a recent hospital admission, lisinopril 20 mg once daily was started for an indefinite period.
A few weeks later, the patient visits his GP for a blood pressure check. The GP looks at the patient's medication overview, but overlooks the fact that lisinopril is already included, due to the large number of medications. Because the blood pressure is still too high, she starts treatment with lisinopril:
- MA: lisinopril 20 mg tablet, 1 tablet once daily, from today
- VV: lisinopril 20 mg tablet; 30 tablets
When the supplier receives the prescription, she sees that there is already an ongoing MA for lisinopril. There are now two MAs for this medicine. The supplier contacts the GP. The GP decides to close her MA and records a stop-MA with the reason ‘Double registration of medication treatment’.
26 Merging building blocks under a single pharmaceutical treatment
A GP has prescribed escitalopram to a patient for an anxiety disorder.
- MA: escitalopram 40 mg tablet, 1 tablet once daily
The patient has not given consent for his medication data to be made available. As a result, when he records his medication use in his PGO, he does not have access to the MA. Consequently, the MGB is recorded in a new MBH.
After some time, the patient informs the GP that he now agrees to making his medication data available. He has also opened his PGO for querying. The GP can now see that building blocks for propranolol exist in two different MBHs.
The patient’s MGB was recorded later than the MA and has the same type of MBH-id. According to the agreements in the Implementation guide migration and hybrid situation V2.0, this would mean that the GP’s MBH should be closed. The EVS also proposes this to the GP. However, the GP decides to maintain her own MA and to close the patient’s MBH instead.
27 Sending renal function value along with new prescription
A patient is prescribed dabigatran for the first time to prevent stroke. The patient has reduced renal function and there is a recent renal function value available in the prescriber’s information system. Based on this renal function value, the prescriber records an MA with a lower dose of dabigatran than the standard dose.
- MA: dabigatran 110 mg capsule, 1 capsule twice daily
- VV: dabigatran 110 mg capsule; 120 capsules
The prescriber sends the prescription (MA + VV) with the laboratory result of the renal function value to the supplier. This is done using the transactions Sending medication prescription and Sending laboratory results.

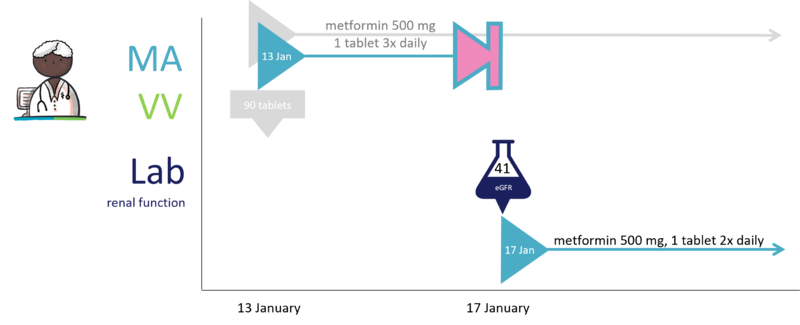
28 Renal function value reason for dosage change
A patient was prescribed metformin on 13 January.
- MA 13 January: metformin 500 mg tablet, 1 tablet 3 times daily
- VV 13 January: metformin 500 mg tablet; 90 tablets
At the same time, the prescriber requested a renal function value test.

On 17 January, the results have come in, showing that renal function was impaired. The prescriber therefore adjusts the dosage.
- MA 17 January: metformin 500 mg tablet, 1 tablet twice daily
The prescriber sends the new prescription (technical stop-MA + new MA) with the laboratory result of the renal function to the supplier. Because the patient had already collected the medication and therefore has sufficient stock, no new VV is sent.
29 Renal function test required due to type of medicine
A patient has cystitis. The GP prescribes a 5-day course of nitrofurantoin, which the patient must start immediately.
- MA: nitrofurantoin 100 mg MR capsule, 1 capsule twice daily, from today, for 5 days
- VV: nitrofurantoin 100 mg MR capsule; 10 capsules
For this medicine, a renal function value (not older than 13 months) must be provided if available. The prescriber has a renal function value for the patient, which does not indicate a need for dose adjustment. The prescriber sends the prescription (MA + VV) with the laboratory results for the renal function value to the supplier.

30 Starting a variable dosing regimen
The prescriber prescribes anticoagulant medication for a patient and refers the patient to the anticoagulation clinic. In the MA, he indicates that the medication should be used according to the dosing schedule of the anticoagulation clinic. The prescriber creates an initial WDS to bridge the period until the start of anticoagulation care.
After the referral, the anticoagulation clinic establishes a dosing schedule that follows the prescriber’s schedule. From that moment on, the anticoagulation clinic creates the subsequent dosing schedules.

31 Interim modification of a variable dosing regimen
A patient is scheduled to undergo a minor procedure. The patient uses anticoagulant medication and must stop taking it several days before the procedure. The current WDS continues until after the day of the procedure. The thrombosis physician creates a new WDS with the dose set to zero for the days preceding the procedure. A technical stop-WDS is created. The MA stays unchanged in the meantime.

32 Stopping medication with a variable dosing regimen
An 88-year-old patient has a history of several falls and frequent bruises. The GP decides, in consultation with the thrombosis specialist, that it is preferable to stop his anticoagulant medication. The GP therefore creates a stop-MA. The stop-MA makes the underlying WDS and TA invalid. For the TA also a stop-TA is recorded.

33 Paper prescription – no more allowed
The Dutch law on electronic data exchange in healthcare (Wet elektronische gegevensuitwisseling in de zorg, Wegiz) states that as of 1 January, 2024, prescriptions on paper only are no longer permitted. Therefore, the contents of this practical example have been removed. There are, however, exceptions conceivable where a paper prescription may still be needed. Examples include:
- foreign patients without a BSN
- the patient's preferred pharmacy is not in the Netherlands.
- network failure
The supplier will then have to make a TA without referral to an MA.
34 Own items (90 million numbers)
A baby born prematurely is discharged from hospital. The public pharmacy receives a prescription for this child for a caffeinated drink 10 mg/ml. This is to stimulate the child’s respiratory centre.
- MA: caffeine drink 10 mg/ml, 1.5 ml once daily (total 15 mg)
The G-standard contains 3 items from 3 suppliers for this. However, the pharmacy has the drink prepared by another supplier. This item cannot therefore be registered as a G-standard product. The pharmacy creates a 90 million number for this. This enables correct declaration, labelling and administration registration. This 90 million number is used when recording the TA.
35 New medication agreement - dispensing the same product
A patient visits the pharmacy to collect her medication. It concerns a new MA for this patient.
- MA: nitrofurantoin 100 mg capsule MR, 1 capsule twice daily, from today, for 5 days
- VV: nitrofurantoin 100 mg capsule MR; 10 capsules
The supplier selects a product based on the MA and factors such as the health insurer’s preference policy and the stock available in the pharmacy. She records a TA and an MVE and dispenses the medication.
- TA: Furabid 100 mg MR capsule, 1 capsule twice daily, from today, for 5 days
- MVE: Furabid 100 mg MR capsule; 10 capsules
36 New medication agreement – different administration agreement
A patient visits the pharmacy to collect her medication. It concerns a new MA for this patient.
- MA: nitrofurantoin 100 mg MR capsule, 1 capsule twice daily, from today, for 5 days
- VV: nitrofurantoin 100 mg MR capsule; 10 capsules
The supplier selects a different product, for example because of the health insurer’s preference policy. She records a TA and an MVE and supplies the medication to the patient.
- TA: Actavis nitrofurantoin 50 mg MR capsule, 1 capsule four times daily, from today, for 5 days
- MVE: Actavis nitrofurantoin 50 mg MR capsule; 20 capsules
37 Proposal medication agreement sent to prescriber for dose adjustment
The supplier receives an MA for gabapentin for a patient.
- MA: gabapentin 600 mg tablet, 1 tablet three times daily
The pharmacy system records that the patient has reduced renal function. When the supplier attempts to record the TA, a medication surveillance alert appears indicating that the maximum dose for this patient is 900 mg per day. The supplier sends a proposal medication agreement (VMA) with an adjusted dose to the prescriber. The prescriber approves the proposal.
The prescriber stops the original MA and records a new MA for a dosage of 300 mg 3 times daily:
- MA: gabapentin 300 mg tablet, 1 tablet three times daily
Because this has a different PRK, the new MA is recorded in a new MBH. The prescriber sends the stop-MA, the new MA and an AVMA back to the supplier, who records a TA in the new MBH :
- TA: gabapentin 300 mg tablet, 1 tablet three times daily
38 Proposal medication agreement sent to prescriber for temporarily stopping medication
A patient is using simvastatin for an indefinite period. A 5-day course of fluconazole has now been prescribed. The supplier records a TA for fluconazole. A notification appears advising that simvastatin should be temporarily stopped due to a potential interaction. The supplier sends a VMA with this recommendation to the prescriber. The prescriber approves the proposal and interrupts the simvastatin for the duration of the fluconazole course.

39 Proposal medication agreement sent to prescriber for adding another medicine
A supplier records a TA for a 55-year-old patient for prednisone.
- TA: prednisone 5 mg tablet, 2 tablets once daily, from today
A notification appears regarding an increased risk of osteoporosis if prednisone is used for longer than 3 months. The patient is already taking calcium and vitamin D but is not using a bisphosphonate. The supplier sends a VMA to the prescriber for alendronic acid tablet 70 mg, once per week. The prescriber approves the proposal and issues an MA for alendronic acid.

40 Long-term medication with repeats in the dispense request
A patient has been using enalapril at the same dose for hypertension for several years. The GP has created an MA for an indefinite period. The TA is also valid for an indefinite period.
- MA: enalapril tablet 10 mg, 1 tablet once daily
The GP records a VV for this every year.
- VV: enalapril tablet 10 mg; 90 tablets
In the VV data element NumberOfRefills, the GP specifies that this should be repeated 3 times.
The patient initially receives 90 tablets. The next time the patient visits the pharmacy, a dispensing is made under the existing agreements; no new VV is required.

41 Long-term medication, supplier makes partial dispensings
A patient has been using enalapril at the same dose for hypertension for several years. The GP has created an MA for an indefinite period. The TA is also valid for an indefinite duration.
- MA: enalapril tablet 10 mg, 1 tablet once daily
The GP issues an annual prescription with a VV for a full year.
- VV: enalapril tablet 10 mg; 360 tablets
The supplier divides this into 4 partial dispensings. The patient initially receives 90 tablets. After that, the patient can receive 90 tablets on three further occasions at the pharmacy; no new VV is required.

42 RequestDate with multiple partial supplies
A GP prescribes enalapril for an indefinite period to a patient with hypertension.
- MA: enalapril 20 mg tablet, 1 tablet once daily
- VV : enalapril 20 mg tablet; 360 tablets
The supplier receives the prescription on 3 February and records a TA:
- TA: enalapril 20 mg tablet, 1 tablet once daily
The patient visits the pharmacy on 6 February to collect the medication. A first medication supply is made under this TA for a period of three months. The RequestDate in the MVE corresponds to the date the prescription was received, the MedicationDispenseDateTime corresponds to the date of dispensing.
The second dispensing is done based on the existing MA, VV, and TA; no new prescription or TA is required. The second MVE receives a new RequestDate and MedicationDispenseDateTime.

43 Patient requests repeat prescription via prescriber
On 30 March, a GP prescribes bisoprolol to a patient as maintenance treatment for stable angina pectoris.
- MA: bisoprolol 5 mg tablet, 1 tablet once daily, from 30 March
- VV: bisoprolol 5 mg tablet; for 3 months
When the patient’s supply runs out, he contacts the GP by phone to request a repeat of this medication. The GP approves the request and creates a new VV, sending it along with the associated MA to the patient’s pharmacy.

44 Patient requests repeat prescription via supplier
On 30 March, a GP prescribes bisoprolol to a patient as maintenance treatment for stable angina pectoris.
- MA: bisoprolol 5 mg tablet, 1 tablet once daily, from 30 March
- VV: bisoprolol 5 mg tablet; for 3 months
When the patient’s supply runs out, he requests a repeat of this medication from the supplier. The supplier sends a VVV to the prescriber. The prescriber approves the proposal and sends an AVVV and a new VV with the associated MA back to the supplier.

45 Proactive repeat prescription by supplier
A patient is registered in the pharmacy’s information system for a proactive repeat of his medication. When the patient requires new medication, the AIS will generate a signal. The supplier then sends a VVV to the prescriber. If the prescriber approves, she sends an AVVV and a new VV with the associated MA back to the supplier.

46 Dispensing a larger quantity than prescribed
The GP prescribes the following for a patient requiring vitamin D:
- MA: colecalciferol 10 micrograms tablet, 1 tablet once daily, from today for 28 days
- VV: colecalciferol 10 micrograms tablet; 28 tablets
The prescription is sent to the supplier.
The supplier only has 30-unit packs available. This means there will be two tablets remaining at the end of the PeriodOfUse, but the intention of the prescription is still met. Because vitamin D at this dose is not prescription-only, a larger pack may be dispensed in this case. For prescription-only medicines, this is not permitted.
A TA is created for 28 days. In the MVE, it is noted that 30 units were supplied, optionally with an explanation for why this differs from the TA.
- TA: colecalciferol 10 micrograms tablet, 1 tablet once daily, from today for 28 days
- MVE: colecalciferol 10 micrograms tablet; 30 tablets
47 Dispensing a smaller quantity than prescribed
A GP prescribes pantoprazole to a patient with stomach complaints:
- MA: pantoprazole 20 mg tablet, 1 tablet once daily, from today, for 30 days
- VV: pantoprazole 20 mg tablet; 30 tablets
The supplier only has 28-tablets packs available and therefore contacts the prescriber. A supply of 28 tablets is dispensed with a TA for 28 days. Both the TA and the MVE now differ from the MA.
- TA: pantoprazole 20 mg tablet, 1 tablet once daily, from today, for 28 days
- MVE: pantoprazole 20 mg tablet; 28 tablets
48 Starting medication in GDS
An elderly patient is prescribed metoprolol on 2 January.
- MA 2 January: metoprolol 100 mg MR tablet (succinate), 1 tablet once daily, from today
- TA 2 January: PCH metoprolol 95 mg MR tablet, 1 tablet once daily, from today
The patient is using so many different medicines that he no longer has a good overview of them.
On 6 February, the prescriber and supplier agree that the patient’s medication will henceforth be provided via GDS. For metoprolol, the process is as follows:
To start GDS, on 7 February the supplier selects a dosing interval for the patient and creates a new TA. A technical stop-TA is also created. The RegistrationDateTime is 7 February. It takes a few days to start GDS, so the startDateTime is 11 February.
- TA 7 February: PCH metoprolol 95 mg MR tablet, 1 tablet once daily in the morning with breakfast, from 11 February
The medication is dispensed via GDS every two weeks from 11 February. In the VV data element DistributionForm of each MVE is indicated that GDS is used.

49 Adding medication to GDS
On 12 October, the GP prescribes dabigatran to a patient for the prevention of CVA and systemic embolism in atrial fibrillation. The patient must start this medication immediately.
- MA: dabigatran etexilate 150 mg capsule, 1 capsule twice daily
The patient receives their medication via GDS. In the data element AdditionalInformation of the MA, the prescriber notes: 'Directly on administration list or in GDS'.
The next medication roll change occurs five days later. The supplier bridges the interim period with a separate supply of the new medicine and creates a TA for this, with endDateTime set to the date of the next medication roll change. She also creates a TA for the GDS supply, with startDateTime set to the date of the next medication roll change.
- TA: CF dabigatran etexilate 150 mg capsule (in strip), 1 capsule twice daily, from 12 October for 5 days
- TA GDS: CF dabigatran etexilate 150 mg capsule (in GDS), from 17 October
50 GDS supplier delivers a different trade product
The supplier creates a (future) TA for a patient on 1 July for metoprolol.
- TTA1 1 July: PCH metoprolol 95 mg MR tablet, 1 tablet once daily, from 4 July
The patient receives this medicine via GDS. On 3 July, however, the GDS supplier delivers a different trade product. Because this has a different HPK, the TA needs to be modified. A technical cancelation-TTA is created since it concerned a future supply.
- TTA2 3 July: Sandoz metoprolol 95 mg MR tablet, 1 tablet once daily, from 4 July
In the data element AdministrationAgreementReasonModificationOrDiscontinuation, the supplier records ‘Drug not available - out of stock’.

51 Supplier changes brand product
A patient has been using metoprolol since 2 January.
- MA 2 January: metoprolol 100 mg MR tablet (succinate), 1 tablet once daily, from today
- TA 2 January: PCH metoprolol 95 mg MR tablet, 1 tablet once daily, from today
After six months, this brand product is no longer available, and a different brand is supplied. The TA must be modified for this.
- TA 3 July: Sandoz metoprolol 95 mg MR tablet, 1 tablet once daily, from today
In the data element AdministrationAgreementReasonModificationOrDiscontinuation, the supplier records: ‘Drug not available - out of stock’.

52 Parallel administration agreements for GDS and non-GDS dispensing
A patient takes 20 mg pantoprazole from the brand Sandoz two to three times daily for stomach complaints.
- TA: Sandoz pantoprazole 20 mg tablet, 1 tablet 2–3 times daily
After some time, it is decided that the patient’s medication will be supplied via GDS. For pantoprazole, this means that the fixed dose of 2 tablets will be supplied in GDS. These are Teva tablets because the GDS supplier does not provide pantoprazole from Sandoz. The third ‘as needed’ tablet continues to be dispensed separately. The supplier modifies the existing TA by recording a technical stop-TA and two new TAs within the same MBH, with the same startDateTime.
- technical stop-TA: Sandoz pantoprazole 20 mg tablet,
AdministrationAgreementStopType'Discontinued' - TA: Sandoz pantoprazole 20 mg tablet, 1 tablet once daily as needed
- TA GDS: Teva pantoprazole 20 mg tablet, 1 tablet twice daily
53 Parallel administration agreements, one of which is modified
The GP prescribes pantoprazole for a patient with dyspeptic symptoms:
- MA: pantoprazole 20 mg tablet, 1 tablet 2–3 times daily
The fixed dose of 2 tablets is supplied via the GDS system and provided by the GDS supplier as Sandoz. The third tablet is for use ‘as needed’ and is dispensed separately as Teva. This results in two parallel TA’s.
At a certain point, the GDS supplier is temporarily unable to provide pantoprazole. The supplier has Teva in stock and modifies the TA for the fixed dose from dispensing in GDS to dispensing in separate packs. Stopping the GDS TA does not affect the ‘as needed’ TA; this continues unchanged.
This results in two parallel TA’s of the same brand, both dispensed separately. However, one contains the fixed dose and the other the ‘as needed’ dose. This way, they remain visible separately on the medication administration record as the fixed dose and the ‘as needed’ dose.
Once pantoprazole can again be supplied via GDS, the supplier will stop the TA for the fixed dose and create a new TA for supply in GDS.
54 Dispensing under someone else’s administration agreement
A patient has been taking olanzapine since being diagnosed with schizophrenia.
- MA: olanzapine 15 mg tablet, 1 tablet once daily
He goes on holiday and notices upon arrival on Friday evening that he has forgotten his medication. He goes to a local out-of-hours GP service and requests tablets for the holiday period. The on-call doctor creates a VV under the existing MA and sends this, together with a copy of the MA, to the out-of-hours pharmacy.
- VV: olanzapine 15 mg tablet; 7 tablets
The supplier at the out-of-hours pharmacy sees the prescription and creates an MVE under the existing TA for the 7 tablets the patient requires, and informs the on-call doctor of this supply. A copy of the original TA is also sent by the supplier.

55 Modifying another person’s administration agreement
A patient is taking pantoprazole from the brand Sandoz for dyspeptic symptoms. During a holiday, he notices that he has forgotten his medication. He attends a local GP practice and requests tablets for the holiday period. The GP sends a VV to the local pharmacy under the existing MA.
The supplier at the local pharmacy finds that they do not have a supply of Sandoz pantoprazole, but only Teva. Consequently, they stop the current TA, start a new TA for Teva pantoprazole, and create an MVE for 28 tablets.
The stop-TA is sent to the pharmacy where the original TA originated. That TA must be resumed in due course.
56 Increasing the dose of GDS medication in a new MBH
A patient receives 1 tablet of losartan 50 mg daily. This has insufficient effect, and the prescriber decides to increase the dose to 100 mg once daily. This is a change at the PRK level. Consequently, the prescriber stops the original MA and records the new MA in a new MBH. In the data element AdditionalInformation she indicates 'Direct on the administration list or in GDS'.
However, the patient still has a few days’ supply of 50 mg tablets in their GDS roll. The supplier decides to supplement the GDS medication with 1 separate losartan 50 mg tablet until the medication roll change.
The supplier records a stop-TA and 3 new TAs:
- stop-TA to stop the original TA as a result of the stop-MA.
- TA for the original 50 mg losartan dose in GDS.
- TA for bridging with separate losartan 50 mg tablets.
- TA for the new 100 mg losartan dose in GDS.
At the next roll change, the increased dose will be supplied in the GDS roll.

57 Increasing the dose of GDS medication within the same MBH
A patient is administered 1 tablet of furosemide 40 mg every day due to oedema. This has insufficient effect and the prescriber decides to increase the dose to 2 tablets once daily. There is no change at PRK level, so this is a regular modification. The prescriber records a new MA within the same MBH. Along with this, a technical stop-MA is created.
The modification must take effect immediately. However, the medication roll change will not take place until several days later. The supplier therefore decides, until the medication roll change, to supplement the GDS medication with separate furosemide 40 mg tablets.
The supplier records a technical stop-TA and 3 new TA’s:
- technical stop-TA to stop the original TA, as a result of the stop-MA.
- TA for the original dosage of furosemide in GDS.
- TA for the bridging period with separate furosemide 40 mg tablets.
- TA for the new dosage of furosemide 40 mg in GDS.
At the next medication roll change, the increased dosage (2 tablets per administration) is supplied in the GDS roll.

58 Increasing the dose of GDS medication with different administration times
A patient is administered 1 tablet of furosemide 40 mg every day at 08:00, due to oedema. This has insufficient effect and the prescriber decides to increase the dose to 1 tablets twice daily, in the morning at 08:00 and in the afternoon at 17:00. There is no change at PRK level, so the new MA and the technical stop-MA are recorded within the same MBH.
The modification must take effect immediately. However, the medication roll change will not take place until several days later. The supplier therefore decides, until the medication roll change, to supplement the GDS medication with separate furosemide 40 mg tablets. The tablet in the GDS roll is still scheduled for 08:00, while the separate tablet will be administered at 17:00 (in data element AdministrationDateTime).
The supplier records a technical stop-TA and 3 new TA’s:
- technical stop-TA to stop the original TA, as a result of the stop-MA
- TA for the original dosage of furosemide in GDS, with administration time 08:00
- TA for the bridging period with separate furosemide 40 mg tablets, with administration time 17:00
- TA for the new dosage of furosemide 40 mg in GDS, with administration times 08:00 and 17:00
At the next medication roll change, the increased dosage (2 tablets per administration) is supplied in the GDS roll.
59 Reducing the dose of GDS medication in a new MBH
A patient receives 1 tablet of losartan 100 mg daily. Due to the patient’s deteriorating renal function, the prescriber decides to reduce the dose to 50 mg once daily. This is a change at the PRK level. Consequently, the prescriber stops the original MA and records the new MA in a new MBH.
The patient still has a few days’ supply of 100 mg tablets in their GDS roll. However, an administrator is not allowed to split tablets. The change must take effect immediately and cannot wait until the medication roll change, which will take place a few days later.
The supplier therefore arranges for the remaining GDS bags to be collected the same day and replaced with a new GDS roll containing the 50 mg tablets. She stops the original TA and records a new TA for the reduced dose.
60 Reducing the dose of GDS medication within the same MBH
A patient is administered 2 tablets of verapamil 240 mg MR every day. The effect is too strong, and the prescriber decides to reduce the dose to 1 tablet once daily. There is no change at PRK level, so the new MA and the associated technical stop-MA are recorded within the same MBH.
The modification must take effect immediately. However, the medication roll change will not occur until several days later. The supplier therefore arranges for the remaining GDS bags to be collected the same day and provide the tablets separately until the next medication roll change.
The supplier records a technical stop-TA and 2 new TA’s:
- technical stop-TA to stop the original TA, as a result of the stop MA
- TA for the bridging period with separate verapamil 240 mg MR tablets
- TA for the new dosage of verapamil 240 mg MR in GDS
At the next medication roll change, the reduced dosage is supplied in the GDS roll.

61 Changing GDS medication to ‘as needed’ medication
A patient uses temazepam for insomnia. The GP has been trying for some time to persuade the patient to stop this sleep medication. The patient now indicates a willingness to try, but wants to have temazepam available for use as needed. The GP changes the dosage from 10 mg temazepam once daily to 10 mg temazepam 'as needed'.
The patient has just received a new two-week medication roll. The GP wants to act on this momentum and decides that the new MA must take effect immediately. The GDS medication must therefore be collected by the pharmacy on the same day.

62 Changing GDS medication to partly GDS and partly ‘as needed’ medication
The GP prescribes diazepam 5 mg twice daily to a patient for anxiety. The effect is too strong, and the prescriber decides to reduce the dose to 1 tablet of diazepam 5 mg once daily in GDS plus 1 tablet ‘as needed’. There is no change at PRK level, so the stop-MA and new MA are recorded within the same MBH.
The modification must take effect immediately. However, the medication roll change will not occur until several days later. The supplier therefore arranges for the remaining GDS bags to be collected the same day and provide the tablets separately until the next medication roll change.
The supplier records a technical stop-TA and 3 new TA’s:
- technical stop-TA to stop the original TA, as a result of the stop-MA
- TA for the bridging period with separate diazepam tablets
- TA for the new dosage of diazepam in GDS
- TA for the new dosage of diazepam as separate tablets
At the next medication roll change, the ‘as needed’ medication is supplied separately, and the reduced regular dose is supplied in the GDS roll.

63 Administering a changed tablet quantity
The GP prescribes pantoprazole to a patient.
- MA: pantoprazole 40 mg tablet, 1 tablet once daily
- TA: Sandoz pantoprazole 40 mg tablet, 1 tablet once daily
After some time, there are supply issues with pantoprazole 40 mg. The pharmacy therefore supplies pantoprazole 20 mg.
- TA: Sandoz pantoprazole 20 mg tablet, 2 tablets once daily
The home care worker sees the new TA in the eTDR, administers 2 tablets, and records the administration.

64 Correcting an administration
The GP prescribes calcium 500 mg, 1 tablet twice daily, for a patient with osteoporosis. The patient receives support from home care to take her medication. The calcium tablets are large, and the patient has difficulty taking both tablets. Today, she initially refuses the second tablet. The home care worker records administration of 1 tablet, with the reason ‘Medication refused’ in MedicationAdministrationReasonForDeviation. A few minutes later, the patient takes the second tablet. The home care worker corrects the record to 2 tablets, with the reason ‘Incorrect registration of medication’.
This can be recorded in the eTDR in two ways:
Correction using a negative administered quantity
In the eTDR, two additional MTDs are recorded: one with a negative AdministeredAmount and one with the correct quantity.

Correction using an additional MTD

65 Correcting administration after the patient spits out a tablet
The GP prescribes calcium 1000 mg, 1 tablet once daily, for a patient with osteoporosis. The patient receives support from home care to take her medication. The calcium tablets are large, and the patient has difficulty swallowing a tablet.
The home care worker administers 1 tablet and records this in the eTDR. A short time later, the patient spits out the tablet and it is not possible to administer it again.
The home care worker corrects the previous record by entering a new MTD with a negative quantity.

66 Medication not administered
A patient is administered 1 tablet of atenolol 50 mg every day. The patient has a lower blood pressure than usual due to fever. The administrator contacts the prescriber. They jointly decide not to administer atenolol on that day. The administrator records an MTD with '0 units' as AdministeredAmount and 'low blood pressure due to fever' in MedicationAdministrationReasonForDeviation.

67 Postponing an administration
A patient is administered 1 tablet of calcium 1000 mg every day. This morning it is not possible to administer the tablet because the patient is experiencing nausea. The administrator records an MTD with '0 units' as AdministeredAmount and 'patient was nauseous' in MedicationAdministrationReasonForDeviation.
For calcium, a fixed administration time is not required. During the afternoon medication round, the administrator visits the patient again and this time the tablet can be administered successfully.

68 Recording separate MTDs per InjectionPatchSite
A patient is administered 50 units of long-acting insulin glargine every evening by a home care worker. Due to the high number of units, this must be administered in two doses of 25 units. The supplier records this in the Comment data element in the TA.
The care worker first administers 25U with InjectionPatchSite 'left upper leg' and then 25U with InjectionPatchSite 'right upper leg' and MedicationAdministrationReasonForDeviation '2 administrations due to 50 units'. Because only one InjectionPatchSite can be recorded in an MTD, two MTDs are required.

69 Reducing dosage on the patient’s initiative
A patient has been prescribed propranolol. The patient is experiencing severe sleep disturbances and has reduced the medication. The patient records an MGB with the changed dosage and the date from which this reduced dose is being taken in startDateTime. The patient indicates that the reason for the change is an adverse effect in ReasonModificationOrDiscontinuationOfUse and specifies in Comment that the adverse effect is insomnia. The patient also notes that the use is not as agreed, resulting in 'No' in the data element AsAgreedIndicator.

70 Stopping medication on the patient’s initiative
Since starting propranolol, a patient has been experiencing insomnia. On 13 August, he decides to stop the medication without informing the prescriber. The patient records 'Discontinued' in MedicationUseStopType and notes in startDateTime the date on which he stopped.
In ReasonModificationOrDiscontinuationOfUse, he records 'Adverse reaction to drug' and specifies in Comment that the adverse effect is insomnia. He sets the AsAgreedIndicator to 'No'. This indicates that he stopped the medication on his own initiative.
71 Patient requests repeat medication from the prescriber via proposal dispense request
A patient uses medication for skin complaints. His supply has run out, and because he is abroad for a few days, he cannot immediately obtain new medication. He records an MGB with MedicationUseStopType 'Suspended' and notes in ReasonModificationOrDiscontinuationOfUse 'Drug not available - out of stock'.
At the same time, he sends a VVV to the prescriber. If the prescriber approves the proposal, a VV is sent to the supplier. The AVVV is then received by the patient.

72 Medication is used as agreed
The patient has been prescribed ferrous fumarate for anaemia.
- MA: ferrous fumarate 100 mg tablet, 1 tablet twice per week, to be taken half an hour before meals
The medication starts on 4 June and has no end date. On 10 June, the GP records the patient’s medication use for the period from 4 to 10 June.

Instructions on filling in the following data elements in the Medication use building block:
| Data element | Value |
|---|---|
| ProductUsed | ferrous fumarate 100 mg tablet |
| UseIndicator | yes |
| AsAgreedIndicator | yes |
| MedicationUseStopType | leave empty |
| startDateTime | 4 June yyyy |
| endDateTime | 10 June yyyy |
| DosingInstructions | as agreed |
73 Medication is used as agreed and patients intends to keep doing so
The patient has been prescribed ferrous fumarate for anaemia.
- MA: ferrous fumarate 100 mg tablet, 1 tablet twice per week, to be taken half an hour before meals
The medication starts on 4 June and has no end date. On 10 June, the GP records the patient’s medication use and their intention to keep using this medication as agreed.

Instructions on filling in the following data elements in the Medication use building block:
| Data element | Value |
|---|---|
| ProductUsed | ferrous fumarate 100 mg tablet |
| UseIndicator | yes |
| AsAgreedIndicator | yes |
| MedicationUseStopType | leave empty |
| startDateTime | 4 June yyyy |
| endDateTime | leave empty |
| DosingInstructions | as agreed |
74 Medication is used but not as agreed
The patient has been prescribed metformin for diabetes.
- MA: metformin 500 mg tablet, 1 tablet three times daily, to be taken with meals
The medication starts on 4 June and has no end date.
During the first week after starting, the patient regularly forgets to take the dose at lunchtime because he is often out during the day. He takes the morning and evening doses as prescribed. He records this in an MGB.

Instructions on filling in the following data elements in the Medication use building block:
| Data element | Value |
|---|---|
| ProductUsed | metformin 500 mg tablet |
| UseIndicator | yes |
| AsAgreedIndicator | no |
| MedicationUseStopType | leave empty |
| startDateTime | 7 June yyyy |
| endDateTime | 11 June yyyy |
| DosingInstructions | 1 tablet two times daily |
75 Medication not used, this is not as agreed
The patient has been prescribed colecalciferol (vitamin D3) for a vitamin D deficiency.
- MA: colecalciferol 800 IU tablet, 1 tablet once daily, to be taken with a meal
The medication starts on 4 June yyyy and has no end date. During a long weekend away, the patient realizes that he forgot to take the medication. Upon returning home, he records that he did not take the medication during this period.

Instructions on filling in the following data elements in the Medication use building block:
| Data element | Value |
|---|---|
| ProductUsed | colecalciferol 800 IU tablet |
| UseIndicator | no |
| AsAgreedIndicator | no |
| MedicationUseStopType | Suspended |
| startDateTime | 20 July yyyy |
| endDateTime | 23 July yyyy |
| DosingInstructions | leave empty |
76 Medication is not used, this is as agreed
The patient has been prescribed ferrous fumarate for anaemia.
- MA: ferrous fumarate 100 mg tablet, 1 tablet twice per week, to be taken half an hour before meals
The medication starts on 4 June yyyy.
On 12 August yyyy, a follow-up check shows that the patient no longer has anaemia, and the doctor decides to stop the medication. At a subsequent consultation, the doctor records during medication verification that the patient has indeed stopped taking this medication from 12 August yyyy.

Instructions on filling in the following data elements in the Medication use building block:
| Data element | Value |
|---|---|
| ProductUsed | ferrous fumarate tablet 100 mg |
| UseIndicator | no |
| AsAgreedIndicator | yes |
| MedicationUseStopType | Discontinued (or leave empty)* |
| startDateTime | 12 August yyyy |
| endDateTime | leave empty |
| DosingInstructions | leave empty |
* When stopping medication in accordance with the MA/TA, it does not matter whether the data element MedicationUseStopType is filled with the value ‘Discontinued’ or left empty. Both situations are processed correctly on the receiving end and indicate the same type of stop.
If the medication is stopped not in accordance with the MA/TA, the data element MedicationUseStopType is expected to be filled with the value ‘Discontinued’.
77 Medication is not used after the PeriodOfUse and this is as agreed
The patient has an infection and is prescribed amoxicillin on 1 September. The medication must be taken for seven days. The PeriodOfUse ends on 8 September yyyy, after which the patient records that he no longer uses the medicine.

Instructions on filling in the following data elements in the Medication use building block:
| Data element | Value |
|---|---|
| ProductUsed | amoxicillin 500 mg dispersible tablet |
| UseIndicator | no |
| AsAgreedIndicator | yes |
| MedicationUseStopType | Discontinued (or leave empty)* |
| startDateTime | 8 September yyyy |
| endDateTime | leave empty |
| DosingInstructions | leave empty |
* When stopping medication in accordance with the MA/TA, it does not matter whether the data element MedicationUseStopType is filled with the value ‘Discontinued’ or left empty. Both situations are processed correctly on the receiving end and indicate the same type of stop.
If the medication is stopped not in accordance with the MA/TA, the data element MedicationUseStopType is expected to be filled with the value ‘Discontinued’.
78 Medication is not used during the PeriodOfUse and this is as agreed
The patient has seasonal hay fever and has been prescribed cetirizine. The medication is intended to be taken as needed when symptoms occur. The patient has not had symptoms for three months and records that he did not use the medication during that period.

Instructions on filling in the following data elements in the Medication use building block:
| Data element | Value |
|---|---|
| ProductUsed | cetirizine 10 mg tablet |
| UseIndicator | no |
| AsAgreedIndicator | yes |
| MedicationUseStopType | Discontinued or Suspended (or leave empty) * |
| startDateTime | 8 August yyyy |
| endDateTime | 8 November yyyy |
| DosingInstructions | leave empty |
* When stopping medication in accordance with the MA/TA, it does not matter whether the data element MedicationUseStopType is filled with the value ‘Discontinued’ or left empty. Both situations are processed correctly on the receiving end and indicate the same type of stop.
If the medication is stopped not in accordance with the MA/TA, the data element MedicationUseStopType is expected to be filled with the value ‘Discontinued’.
79 Medication is used but unknown whether this is as agreed
The patient has influenza and uses paracetamol 500 mg for muscle pain, purchased from the local drugstore. He has been taking four to six tablets daily for four days and expects to continue this for approximately three more days.
On 12 August yyyy the patient records this as medication use.

Instructions on filling in the following data elements in the Medication use building block:
| Data element | Value |
|---|---|
| ProductUsed | paracetamol 500 mg tablet |
| UseIndicator | yes |
| AsAgreedIndicator | leave empty |
| MedicationUseStopType | leave empty |
| startDateTime | 8 August yyyy |
| DurationOfUse | 7 days |
| DosingInstructions | 4 to 6 tablets per day |
80 Correcting inadvertently outstanding medication (‘orphan’) during medication verification
A patient was admitted to hospital for acute treatment. Several medicines were prescribed during the admission. A medicine that was intended to be temporary was accidentally not stopped upon discharge. The corresponding MA was recorded without an endDateTime. This MA is therefore an ‘orphan’: an incorrectly active building block.
A few weeks after discharge, the patient visits their GP for a check-up. During medication verification, the GP notices the incorrectly outstanding MA and records a stop-MA for this medication. The stop-MA is made available and sent to the prescribing specialist.
81 Medication for single use without a specific intake time
A child has croup. The GP prescribes a single 5 ml dose of dexamethasone oral solution (1 mg/ml). Because this is a single dose and the time of day is irrelevant, the GP indicates this by entering a startDateTime and endDateTime with only the relevant date in the PeriodOfUse.

82 DurationOfUse in the MVE for non-daily GDS medication
A patient takes oral ferrous fumarate 200 mg three times per week for chronic iron-deficiency anaemia. The supplier records a TA for this and supplies the medication via GDS.
- TA: CF ferrous fumarate 200 mg tablet, 1 tablet three times per week (on Mon-Wed-Fri)
- MVE: CF ferrous fumarate 200 mg tablet; 3 tablets; MedicationDispenseDateTime 8 January yyyy, DurationOfUse 7 days
Although the last dose of the week occurs on day 5, a DurationOfUse of 7 days is recorded in the MVE. This ensures that the medication is only supplied once per week, preventing early intake outside the prescribed weekly schedule.

83 DurationOfUse in the MVE when bridging the period until medication is included in GDS
A patient receives long-term medication via GDS, supplied in fourteen-day cycles. On day 4 of the current GDS roll (8 January), the GP prescribes ferrous fumarate 200 mg to be taken orally twice per week for chronic iron-deficiency anaemia. Because the current GDS roll has already been issued, the new medication cannot be included in this cycle.
The supplier records a TA and provides the ferrous fumarate temporarily as separate tablets for a period of ten days, allowing the patient to start treatment immediately.
- TA: CF ferrous fumarate 200 mg tablet (in strip), 1 tablet twice per week (on Mon-Thu), starting immediately for 11 days
- MVE: CF ferrous fumarate 200 mg tablet; 3 tablets; MedicationDispenseDateTime 8 January yyyy, DurationOfUse 11 days
At the next GDS delivery, the ferrous fumarate is included in the GDS roll, and an additional TA is recorded to align with the existing two-week GDS cycle.
- TA: CF ferrous fumarate 200 mg tablet (in GDS), 1 tablet twice per week (on Mon-Thu), starting on day 12
- MVE: CF ferrous fumarate 200 mg tablet; 3 tablets; MedicationDispenseDateTime 19 January yyyy, DurationOfUse 14 days
84 Dispensing after approval of proposal medication agreement for additional medication
The supplier receives a prescription for oxycodone for a patient.
- MA: oxycodone 10 mg tablet, 1 tablet twice daily
- TA: oxycodone 10 mg tablet, 1 tablet twice daily
The prescriber has forgotten to prescribe a laxative. The supplier sends a VMA for magnesium hydroxide to the prescriber and waits for the prescriber’s approval.
- VMA: magnesium hydroxide 724 mg tablet, 4–10 tablets once daily; Proposal comment: Please initiate a laxative alongside the opioid to prevent and treat opioid-induced constipation.

The prescriber agrees to prescribe a laxative but opts for a medicine with a different PRK. The prescriber sends an AVMA with the decision ‘Rejected’ and a new prescription for macrogol back to the supplier.
- AVMA: Proposal for magnesium hydroxide 724 mg tablet rejected; Reply comment: Macrogol is first choice according to the guidelines and is therefore preferred over magnesium.
- MA: macrogol/electrolytes powder for oral solution, 1 sachet 1–2 times daily
- VV: macrogol/electrolytes powder for oral solution; 28 sachets
As this concerns a different PRK, the MA for macrogol is recorded in a new MBH.
The supplier notes that the proposed medicine has been rejected by the prescriber and that the latter has prescribed a different medicine. The supplier records a TA and MVE in the MBH of the MA for macrogol.

85 Dispensing before approval of proposal medication agreement for additional medication
The supplier receives a prescription for oxycodone for a patient.
- MA: oxycodone 10 mg tablet, 1 tablet twice daily
The prescriber has forgotten to prescribe a laxative. The supplier decides to dispense the laxative macrogol. To do so, she records a TA and MVE in a new MBH and sends this to the prescriber via a VMA.
- VMA: macrogol/electrolytes powder for oral solution, 1 sachet 1–2 times daily; Proposal comment: Request for prescription in accordance with dispensed laxative.
- TA: macrogol/electrolytes powder for oral solution, 1 sachet 1–2 times daily
- MVE: macrogol/electrolytes powder for oral solution; 28 units

The prescriber agrees with the proposal and sends an AVMA with the decision ‘Accepted’ back to the supplier, as well as a new MA and VV for macrogol in the same MBH as the TA and MVE. In the new MA, the PeriodOfUse is copied in full from the TA because dispensing has already occurred.
- AVMA: proposal for macrogol/electrolytes powder for oral solution accepted
- MA: macrogol/electrolytes powder for oral solution, 1 sachet 1–2 times daily
- VV: macrogol/electrolytes powder for oral solution; 28 sachets
The supplier receives the AVMA and the new MA and VV. As the new MA has made the original TA invalid, the TA is modified with a technical stop-TA and a new TA.
- technical stop-TA: macrogol/electrolytes powder for oral solution; AdministrationAgreementStopType ‘Discontinued’
- TA: macrogol/electrolytes powder for oral solution, 1 sachet 1–2 times daily
86 Dispensing before approval of proposal medication agreement for replacement medication
The supplier receives a prescription for coal tar/levomenthol (Denorex) shampoo for a patient.
- MA: coal tar/levomenthol shampoo 75/15 mg/g, every 2 days during the first 10 days. After this, apply at least 2 or 3 times per week until the result allows less frequent treatment.
The Denorex shampoo is out of stock. The supplier replaces it with a shampoo containing ketoconazole. The ketoconazole shampoo is dispensed under the MBH of the existing MA. The supplier records a TA and MVE and sends these to the prescriber. She also includes a VMA to ensure that the TA and MA are aligned.
- TA: ketoconazole shampoo 20 mg/g, twice per week
- VMA: ketoconazole shampoo 20 mg/g, twice per week; Proposal comment: Denorex not in stock, therefore an alternative has been provided.
- MVE: ketoconazole shampoo 20 mg/g; 100 ml

The prescriber agrees to the proposal and sends an AVMA with the decision ‘Accepted’ to the supplier. She discontinues the Denorex shampoo with a stop-MA and records a new MA with VV for the ketoconazole shampoo in a new MBH.
The stop-MA is necessary to prevent duplicate medication. In the new MA, the PeriodOfUse is copied in full from the TA as the shampoo has already been dispensed.
- AVMA: proposal for ketoconazole shampoo 20 mg/g accepted
- stop-MA: coal tar/levomenthol shampoo 75/15 mg/g; MedicationAgreementStopType ‘Discontinued’
- MA: ketoconazole shampoo 20 mg/g, twice per week
The supplier sees that the proposal has been accepted and receives the stop-MA and the new presciption. She records a stop-TA and a new TA and sends these to the prescriber.
- stop-TA: ketoconazole shampoo 20 mg/g; AdministratrationAgreementStopType ‘Discontinued’
- TA: ketoconazole shampoo 20 mg/g, twice per week
87 Document history
| Version | Date | Description |
|---|---|---|
| 9 3.0.0-rc.2 | March 2026 | for all changes see: *Releasenotes |

























